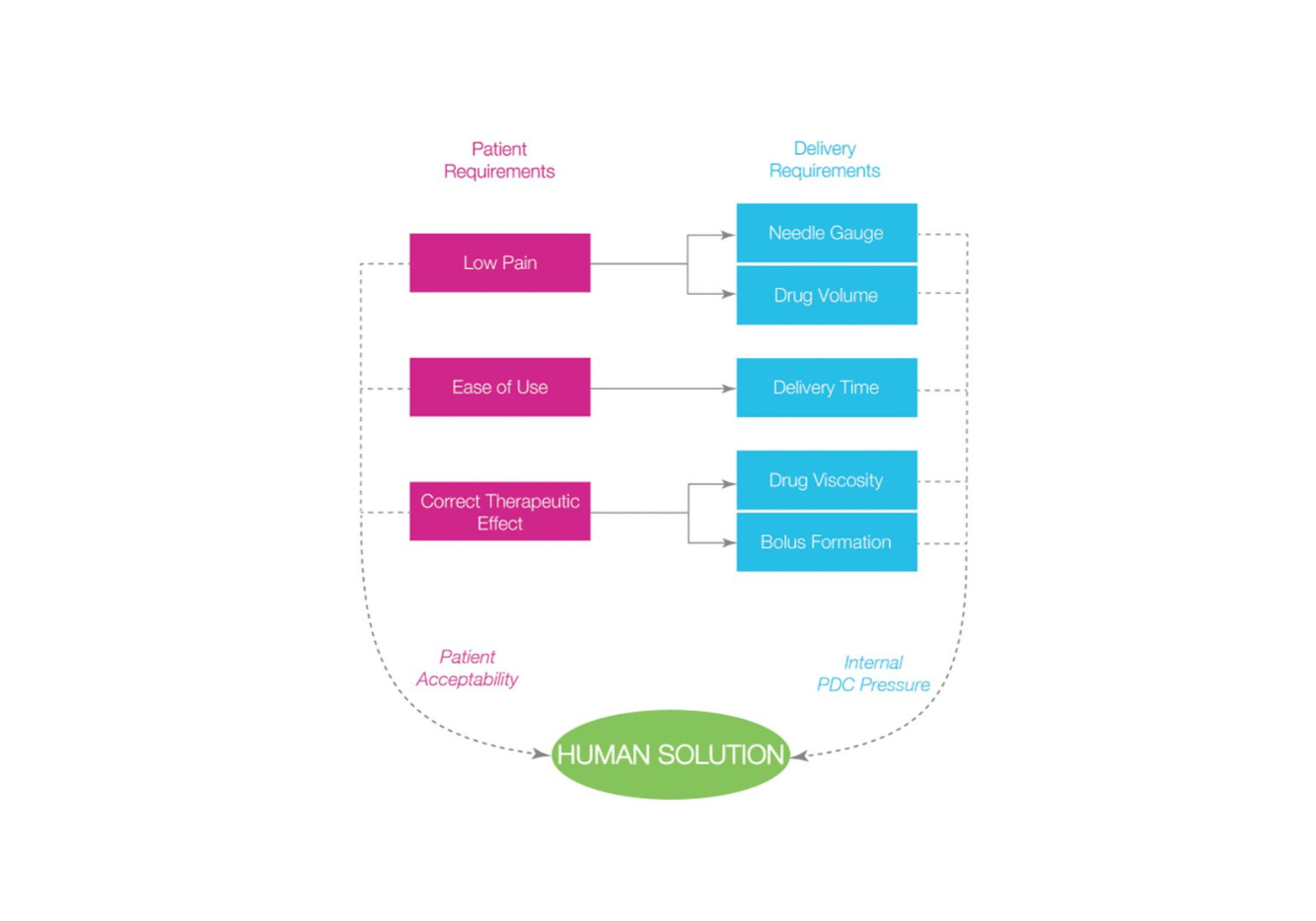
Combining Human Needs With High Viscosity Formulations
February 14, 2025
Join us at the Life Science Forum–Costa Rica June 4, 2026
SMC provides flexible contract manufacturing services to support the development, launch, and scale-up of disposable and point-of-care (POC) diagnostic kits and devices. From self-administered DNA test kits to sepsis diagnostics and battlefield-ready blood tests, our expertise spans a wide range of critical applications.
With experienced teams, robust in-house capabilities, and a global supply chain, SMC ensures precision and efficiency at every stage of diagnostic device production. Our expertise in both high- and low-volume manufacturing allows for seamless scale-up, while FDA-registered, ISO 13485-certified facilities worldwide deliver consistent quality. Trusted by leading diagnostics companies, we are a partner in bringing innovative solutions to market quickly and reliably.
SMC is a dedicated contract manufacturer for the medical market, providing custom contract manufacturing tailored to the unique requirements of diagnostics. All of our global facilities are FDA-registered and ISO 13485-certified, ensuring compliance and consistency at every stage.
From design and engineering to molding, assembly, automation, and final packaging, we deliver a seamless path from concept to commercialization. By uniting these capabilities under one roof, we simplify complex production challenges, accelerate time-to-market, and ensure reliable, high-quality outcomes for both new innovations and established diagnostic product lines.

Specialized in high-precision tooling design and manufacturing for complex applications, ensuring performance and durability.

Specialized injection molding capabilities in silicone, thermoplastics , and micro/miniature components, supported by in-house tooling.

In-house automation and ISO Class 7 & 8 clean rooms streamline assembly, reduce risk, and optimize throughput.

Advance your diagnostic innovation from concept through production with our dedicated design center.
As a privately owned, global contract manufacturer, SMC has partnered with many of the world’s leading medtech and diagnostics OEMs for more than three decades. Customers trust us to deliver efficient scale-up and mitigate risk through robust quality management, regulatory compliance, and resource planning systems designed for consistency and reliability.
The need for medical manufacturing methods that support adaptable, scalable solutions has become more crucial than ever in the diagnostic space. As the technologies associated with creating a lab-on-a-chip become more complex, so does the process of bringing the assay to a fully commercialized product. Our customers rely on us for speed to market with high-quality results.

A single point of contact ensuring seamless communication, performance, and compliance throughout the project, minimizing risk and meeting cost, volume, and regulatory requirements.

Specialists in designing and manufacturing microfluidic cartridges and cassettes, ensuring precise execution of complex POC consumables from concept to commercialization.

Specialized in optimizing global supply chains, ensuring seamless sourcing, logistics, and inventory management, minimizing risks, reducing costs, and streamlining product flow.

SMC is FDA-registered, MedAccred accredited, and backed by an ISO 13485-certified Quality Management System (QMS) for compliance and reliability.
Yes, SMC is a global organization with 11 locations – United Kingdom, India, Costa Rica and 9 located in the United States (California, Ohio, Wisconsin (4), Massachusetts, North Carolina).
All SMC facilities are designed to produce high-volume medical devices, but each location has unique specialties. For example: SMC Somerset specializes in silicone manufacturing. North Carolina serves as our dedicated fill-finish location. Our facilities are strategically located to provide convenient access and support to our customers. To determine which facility is best suited for your project, we recommend reaching out to us for personalized guidance. Contact us for more information.
All of SMC manufacturing fall in either Medical Device, Diagnostics or the Pharmaceutical Markets. We do not manufacture anything in other markets.
At SMC, we stand out among other CDMOs (Contract Development and Manufacturing Organizations) because of our unique approach and capabilities. Not only are we a privately held company with an actively engaged management team, but we also specialize exclusively in medical manufacturing, serving the medical device, diagnostic, and pharmaceutical markets.
Our end-to-end manufacturing solutions cover everything from design to production, including fill-finish capabilities. With subject matter experts (SMEs) available every step of the way, we ensure a seamless process tailored to your needs.
What truly sets SMC apart is our proprietary auto-injector technologies, which we offer exclusively to our customers.
At SMC, the sooner we can get involved, the better! However, we’re happy to assist at any stage of your project. Whether you’re starting with a simple concept—like a napkin sketch—or transferring a fully developed program through a lift-and-shift process, we’re here to help.
It’s always best to engage your Contract Manufacturer (CM) as early as possible during your program’s planning phase. Early collaboration allows us to partner with you effectively and thoroughly review the manufacturing aspects of your project for optimal success.
At SMC, the collaborative supply chain teams, led by our Chief Supply Chain Officer, employ best-in-class techniques to ensure customer success on a global scale. The SMC-approved supplier list includes multiple alternatives for all major commodities, providing diverse sourcing options to mitigate risk. We collaborate closely with internal stakeholders, including finance, engineering, operations, and legal teams, to ensure alignment and support for procurement strategies tailored to address customer concerns and needs. By building and nurturing strong relationships with key suppliers and partners, we leverage strategic advantages that foster innovation, enhance pricing power, and provide flexibility in production and transportation. Key performance indicators (KPIs) are utilized to measure the effectiveness of procurement strategies and operations, driving continuous improvement in supply base processes, systems, and practices. Our approach anticipates and mitigates risks by accounting for economic, geopolitical, and environmental events that may impact procurement and supply chain operations. Additionally, we achieve buying power through a combination of concentrated spending with key suppliers, independent pricing and minimum order quantity (MOQ) negotiations, and the use of purchasing data and forecast extrapolations.
Our success in managing potential supply risk is rooted in our focused approach across several key areas. We maintain a strong core of senior technical and commercial leaders within SMC, ensuring that experienced leadership drives risk management efforts. Identifying supply risk areas and implementing mitigation strategies is a core responsibility of our senior management and their teams. To stay ahead of our customers’ capacity needs, we pre-invest in infrastructure, ensuring preparedness and adaptability. Consistent technology investments enable us to mitigate risks related to both obsolescence and security. Additionally, we provide safety stock of both raw materials and finished goods to support our customers’ needs. Where applicable, we employ dual sourcing for critical components to enhance supply chain resilience. Lastly, site-specific business continuity plans allow us to respond quickly and effectively to unexpected events, minimizing potential disruptions.
Yes, all SMC Facilities are ISO 13485 Certified and FDA registered. You Can find all of our certifications here.
Our contract manufacturing capabilities, focused on single-use and disposable medical devices, include innovative autoinjector platforms designed for subcutaneous or intramuscular injection. These platforms can be customized to your drug formulation.