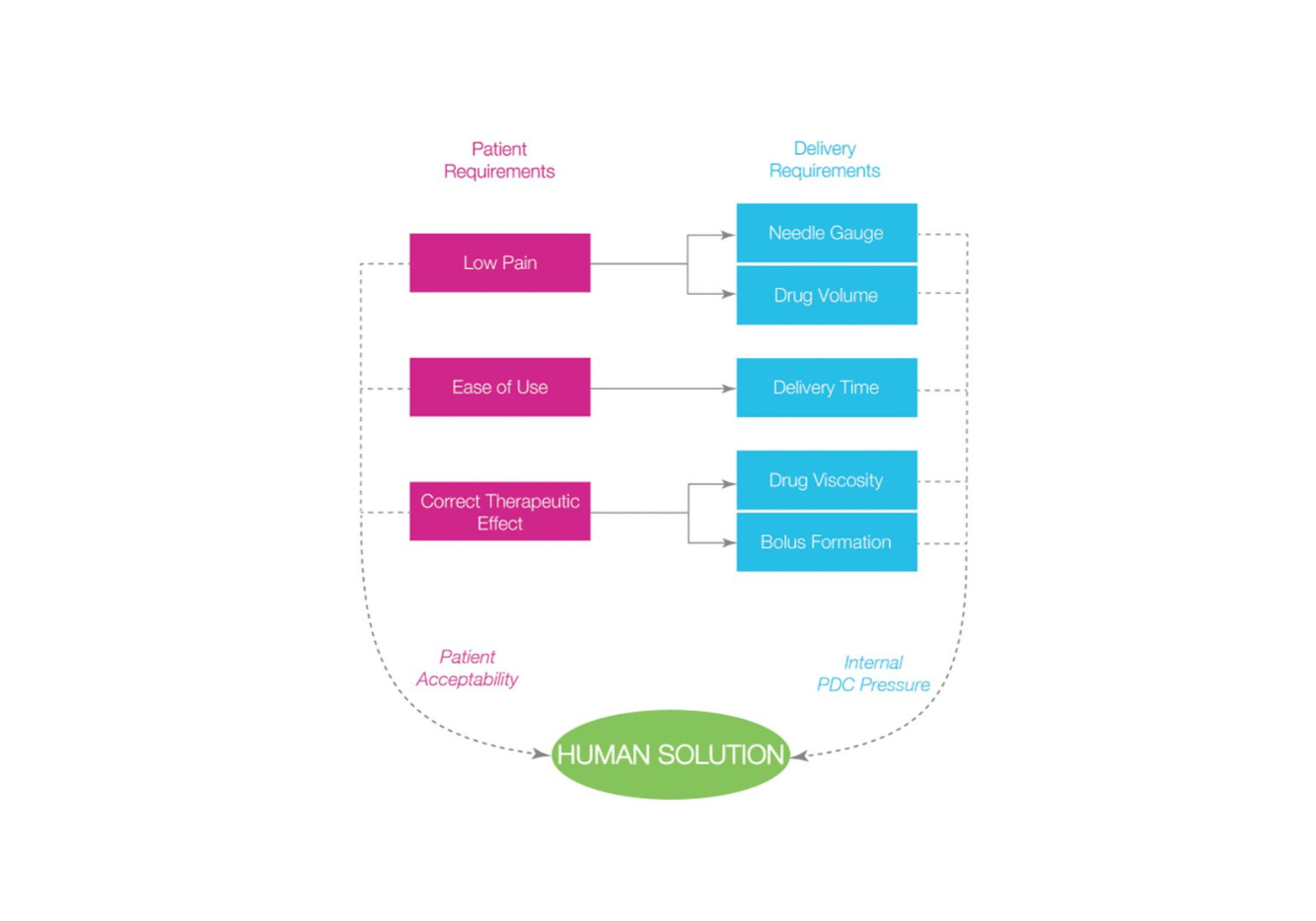
Combining Human Needs With High Viscosity Formulations
February 14, 2025
Within the injectable drug landscape, the availability and usage of high viscosity (HV) drugs is growing, often driven by developments…
Read More
Join us at the American Bio Summit April 14-15, 2026